Aldehydes and Ketones
Next Lesson
Summary
The content provides an in-depth exploration of aldehydes and ketones, focusing on their structure, naming conventions, and physical properties, essential for MCAT Organic Chemistry 2 preparation.
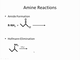
- Aldehydes are characterized by a carbonyl group bonded directly to a hydrogen, with naming conventions including replacing 'e' in alkanes with 'al' or using 'formyl' as a prefix.
- Ketones share the carbonyl functionality but are distinguished by having two alkyl groups attached to the carbonyl carbon, with naming involving replacing 'e' with 'one' or using 'oxo' as a prefix.
- Both aldehydes and ketones exhibit increased polarity, higher boiling points, and greater solubility in water compared to alkanes due to the carbonyl group, with these properties diminishing as the chain length increases.
- The carbonyl carbon in both aldehydes and ketones is slightly positive, making it a target for nucleophilic attack.
Chapters
00:00
Introduction to Aldehydes
01:43
Physical Properties of Aldehydes and Ketones
05:21
Chemical Reactivity and Functional Significance